 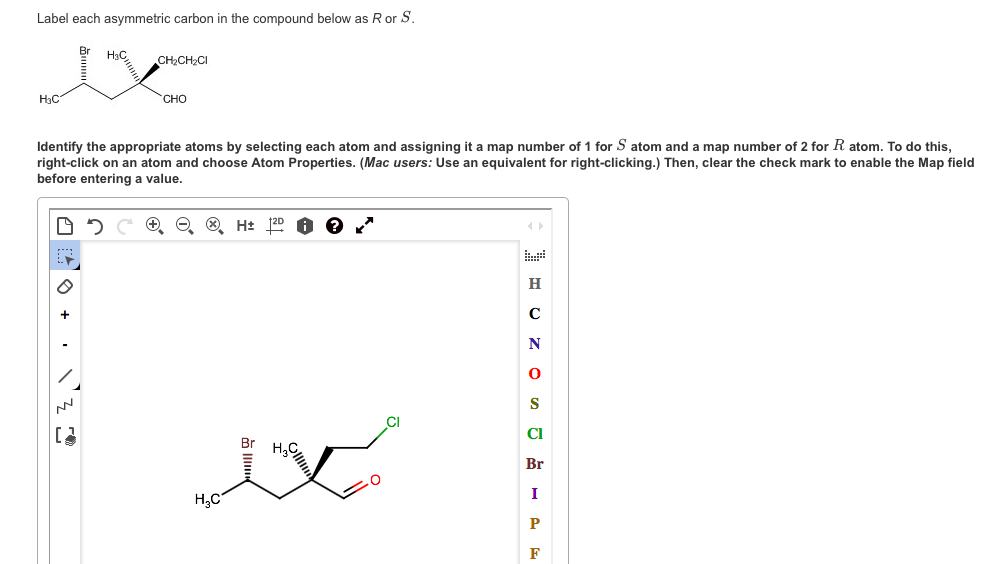 
So, in this structure, there are two chiral centers i.e., the 1st carbon atom and the 2nd carbon atom.ġ-choloro[pentane does not contain any asymmetric carbon atom, Thus it is not chiral compound. So, in the ring structure, we have to find which carbon atom is attached to four different compounds or elements. Can rings be chiral?Ĭhiral center means the carbon atom in the system that has all the different substituents. Phosphate triesters are chiral if the all four substituent groups are different (including the carbonyl oxygen). In order to investigate the stereochemistry of reactions at the phosphate center, 17O and 18O isotopes of oxygen (the ‘normal’ isotope is 16O) can be incorporated to create chiral phosphate groups. Illustration showing how a chiral sample can rotate plane-polarized light. 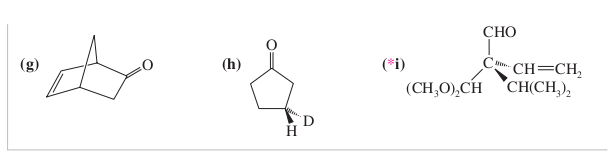
Why chiral carbons are optically active?Īs stated, chiral molecules are optically active, which implies that when a beam of plane-polarized light passes through a chiral molecule, it interacts with the molecule in such a way that the angle of the plane of oscillation rotates. Yes, a compound can be chiral even though it has no chirality centres. The four chiral centres (with their attached groups) are: C2 (OH, C1, C3, H) C3 (OH, C2, C4, H) C4 (OH, C3, C5, H) Can chiral be center without chiral?ġ Answer. The main difference between chiral and achiral is that the mirror image of a chiral is non-superimposable whereas the mirror image of an achiral is superimposable. Main Difference – Chiral vs Achiral A chiral carbon is an asymmetric carbon atom present in a compound. Enantiomers also have chiral centers in the molecules, but not all molecular stereoisomers are mutually enantiomers. What is the difference between chiral and enantiomers?Ĭhiral describes an atom that has four separate groups attached to it, while enantiomers describe the two stereoisomer relations. 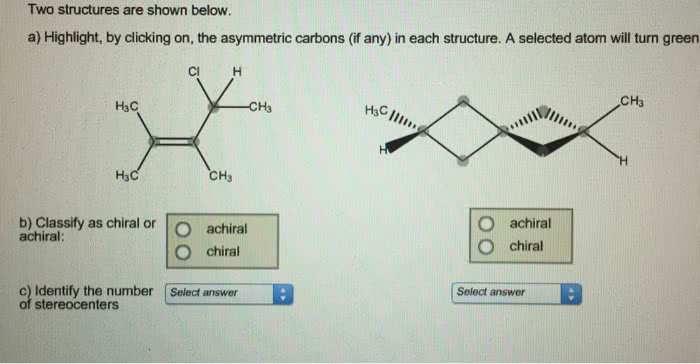
Molecules whose mirror image is non-superimposable over them are known as chiral. The left hand is a non-superimposable mirror image of the right hand no matter how the two hands are oriented, it is impossible for all the major features of both hands to coincide across all axes. Human hands are perhaps the most recognized example of chirality. Achiral molecules are symmetric at all the centres. Chiral molecules are asymmetric at one or more centres. Achiral molecules are superimposable mirror images of each other. What is difference between chiral and achiral?Ĭhiral molecules are non-superimposable mirror images of each other. Given this, we find that there are three chiral carbons. We can immediately eliminate any carbons that are involved in double bonds, or that have two hydrogens attached. The key to finding chiral carbons is to look for carbons that are attached to four different substituents. The lack of a plane of symmetry makes the carbon chiral. What makes a carbon chiral?Ī carbon atom that is bonded to four different substituents loses all symmetry, and is often referred to as an asymmetric carbon. Likewise, gloves and shoes come in pairs, a right and a left. Thus, one can buy right or left-handed golf clubs and scissors. What is chiral and examples?Ĭhiral objects have a “handedness”, for example, golf clubs, scissors, shoes and a corkscrew. A chiral molecule that contains a single stereogenic center may exist in an enantiopure form or as a mixture of enantiomers in varying ratios. The word chirality is used as a term to describe a molecule whose enantiomers are non-superimposable on their mirror images. 24 Why is chirality important in chemistry? What does chiral mean in chemistry? 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
